Background
Glioblastoma multiforme represents approximately half of the cases of central nervous system malignant tumors. The median age at diagnosis is 64 years, and the incidence was reported to be approximately 3 cases in 100 000 people. Genetic conditions, such as Li Fraumeni and Lynch Syndrome, are associated with a higher risk of glioblastoma occurrence. Previous therapeutic irradiation, decreased susceptibility to allergies, impaired immune response and male sex were also associated with this disease. It is one of the most aggressive types of cancer, and only about 1 in 5 patients survive in the second year after diagnosis [1] [2] [3].
Current Clinical Practice
The standard of care treatment consists of surgery, adjuvant radiotherapy, and chemotherapy with Temozolomide (concurrent and adjuvant).
Surgery is performed whenever possible, the complete resection of the tumor offering the chance of longer progression free survival. Adjuvant treatment decisions are influenced by the patient’s age, performance status, and other prognostic factors, like MGMT promoter methylation.
Radiotherapy is usually delivered using conventional fractionation (59.4-60 Gy/30-33 fr). A slightly lower dose is used when the tumor volume is very large or if there is brainstem/spinal cord involvement or close proximity. Another possibility is to irradiate the whole volume with a dose of 45-50.4 Gy and adding a boost up to the total dose mentioned above, on the area at higher risk for microscopic disease persistence. For elderly patients or those with a poor performance status, hypofractionated radiotherapy is given over 2-4 weeks [4].
Alternating Electric Field Therapy and Carmustine Wafers inserted at the time of surgery are newer treatment options, but their impact on the outcomes is low. New systemic treatments, including targeted therapies and immunotherapy, have not shown significant benefits, either.
Since virtually all tumors relapse, other solutions were researched. Currently, over 100 therapies are under evaluation for glioblastomas. The NCCN guidelines recommend the enrollment of patients in clinical trials as a preferred option, in order to increase the chances of finding a more efficient treatment. However, only around 10% of eligible patients are ever enrolled in these studies.
New Studies
A recent meta-analysis of fractionation schedules concluded that even limited dose escalation can be expected to have a clinical benefit for the treatment outcome of glioblastoma patients[5]. At the same time, a retrospective analysis on 171 patients treated in a Danish center suggested that all patients with unifocal disease, and those with multifocal disease who have undergone a partial or complete resection, are candidates for radiation dose-escalation [6].
NRG phase II trial preliminary results showed that the addition of re-irradiation improved progression-free survival when comparing bevacizumab alone in patients with recurrent glioblastomas [7]. Reirradiation is possible at relapse if the appropriate techniques are used, in order to keep the dose to the organs at risk within safe limits. One possibility is to use highly conformal techniques, like SRS.
Another type of irradiation that spares the surrounding tissues is intraoperative radiotherapy (IORT). Both techniques deliver high doses to a low volume: in one fraction (IORT) or up to five fractions (SRS). A phase I/II trial, INTRAGO, concluded that adding IORT to standard radiochemotherapy in newly diagnosed glioblastoma patients was associated with manageable toxicity [8]. Another pooled analysis of 51 patients from 3 countries suggested that adding IORT to standard treatment improved efficacy, when comparing the outcomes with the historical data, without increasing the toxicity [9].
A Challenge: Delineation of Organs at Risk
Precise delineation is crucial for quality and safe treatment delivery, so contouring is reviewed in clinical trials. In 2012, an EORTC trial verified 57 datasets of patients who received irradiation for low-grade gliomas, from 48 institutions. It was observed that OAR contours were missing in 25% of cases on average, and up to one-third of those present, were incorrectly drawn [10]. Another study, from 2018, reported results from retrospective individual cases of anaplastic gliomas. A significant amount of acceptable variations were related to inconsistent OAR outlining. However, if some OAR contours were missing or were anatomically incorrect, the overall delineation was classified as acceptable variation (except for the chiasm, optic nerve or brainstem, which led to classification as unacceptable variation) [11].
Another interesting study from France compared the contours of the cranial OARs made on the same scan by participants from 16 centers and compared them with one delineation set made by two experts in neuro-oncology. They concluded that for the smaller OARs, the agreement decreases. Kappa Index value is 1 for perfect agreement, and it was found to be 0.8 for the parotid and the brainstem, but only 0.5 for the cochlea and 0.3 for the optic chiasm [12].
Glioblastoma is an aggressive disease, for which further treatment improvement is needed. Increasing the radiotherapy dose is promising, but precise irradiation is mandatory to keep the surrounding organs in a safe zone. Accurate and reproducible contouring is necessary to improve the therapeutic ratio and harmonize treatment delivery in clinical trials.
A solution: Guideline-based Brain Model for OARs

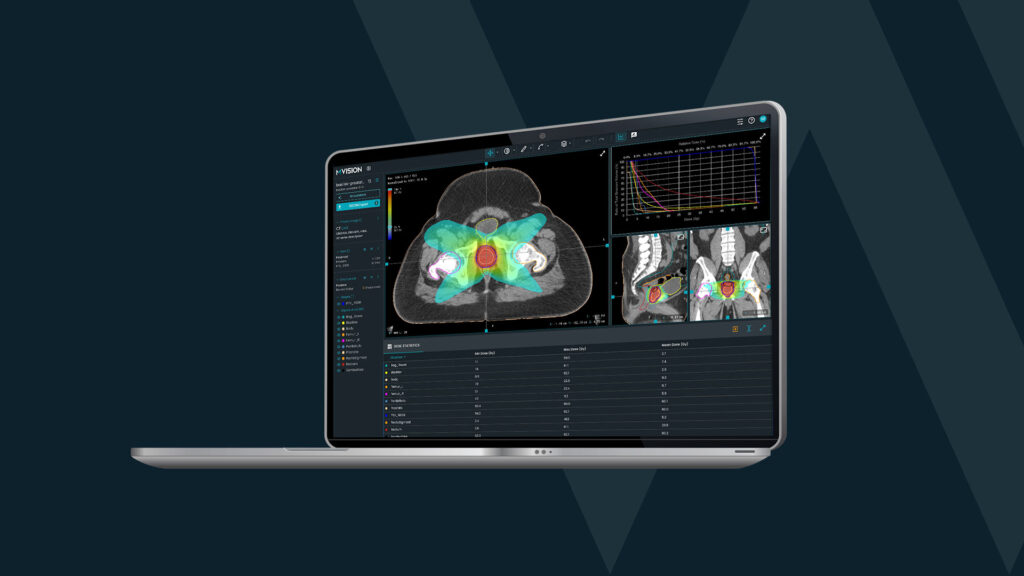
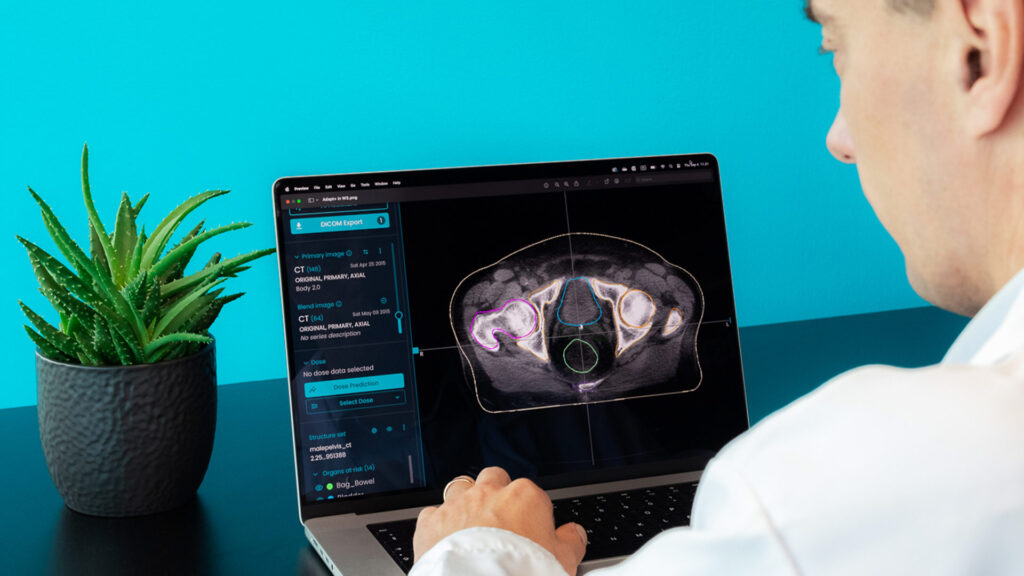
MVision AI’s guideline-based segmentation solution includes Brain CT and MR models. These models are trained with CT and MRI scans and they have the ability to improve standardization and increase consistency when treating brain tumors with conventional fractionation, hypofractionation, and SRS. Both brain models are CE marked and available. The CT model is also available in the US and contains 21 structures, while the MR model supports 28 ROIs for both native and contrast enhanced T1 sequences.
Book a demo to explore GBS™ for your clinic.
MVision AI is active on LinkedIn, Twitter, Facebook, and Instagram. Follow us on social media to stay up-to-date on the latest MVision AI news.
Are you eager to learn more? We welcome you to contact MVision AI:
c/o Terkko Health Hub, Haartmaninkatu 4, 00290 Helsinki, Finland.
Tel: +358 (0) 40 5489 229
Email: info@mvision.ai
For media inquiries:
+358 40 500 7915; pr@mvision.ai
Sources
[1] Grochans, Szymon et al. “Epidemiology of Glioblastoma Multiforme-Literature Review.” Cancers vol. 14,10 2412. 13 May. 2022, doi:10.3390/cancers14102412
[2] Wen PY et al. Glioblastoma in adults: a Society for Neuro-Oncology (SNO) and European Society of Neuro-Oncology (EANO) consensus review on current management and future directions. Neuro Oncol. 2020 Aug 17;22(8):1073-1113. doi: 10.1093/neuonc/noaa106.
[3] https://www.aans.org/en/Patients/Neurosurgical-Conditions-and-Treatments/Glioblastoma-Multiforme
[4] https://www.nccn.org/professionals/physician_gls/pdf/cns.pdf
[5] https://www.estro.org/Congresses/ESTRO-2022/648/cns/10690/metaanalysisoffractionationschedulestohypothesizea
[6] https://www.estro.org/Congresses/ESTRO-2022/598/18-cns/10230/radiationdose-escalationforglioblastoma-whomaybene
[7] Tsien, Christina, et al. “ACTR-32. NRG ONCOLOGY RTOG 1205: randomized phase II trial of concurrent bevacizumab and re-irradiation vs. bevacizumab alone as treatment for recurrent glioblastoma.” Neuro-Oncology 21.Suppl 6 (2019): vi20.
[8] Giordano FA et al. Intraoperative Radiotherapy in Newly Diagnosed Glioblastoma (INTRAGO): An Open-Label, Dose-Escalation Phase I/II Trial. Neurosurgery. 2019 Jan 1;84(1):41-49. doi: 10.1093/neuros/nyy018. PMID: 29528443.
[9] Sarria GR et al. Intraoperative radiotherapy for glioblastoma: an international pooled analysis. Radiother Oncol. 2020 Jan;142:162-167. doi: 10.1016/j.radonc.2019.09.023. Epub 2019 Oct 16. PMID: 31629553.
[10] Fairchild, Alysa et al. Quality assurance in the EORTC 22033–26033/CE5 phase III randomized trial for low grade glioma: The digital individual case review Radiotherapy and Oncology, 2012, Volume 103, Issue 3, 287 – 292
[11] Abrunhosa-Branquinho, André N. et al.Radiotherapy quality assurance for the RTOG 0834/EORTC 26053-22054/NCIC CTG CEC.1/CATNON intergroup trial “concurrent and adjuvant temozolomide chemotherapy in newly diagnosed non-1p/19q deleted anaplastic glioma”: Individual case review analysis. Radiotherapy and Oncology, 2018, Volume 127, Issue 2, 292 – 298
[12] Vogin G, Hettal L, Bartau C, Thariat J, Claeys MV, Peyraga G, Retif P, Schick U, Antoni D, Bodgal Z, Dhermain F, Feuvret L. Cranial organs at risk delineation: heterogenous practices in radiotherapy planning. Radiat Oncol. 2021 Feb 4;16(1):26. doi: 10.1186/s13014-021-01756-y. PMID: 33541394; PMCID: PMC7863275.